Blood pressure drug recalled due to cancer risk
The recall encompasses approximately 580,000 bottles of prazosin hydrochloride which contain nitrosamine impurities considered potentially carcinogenic
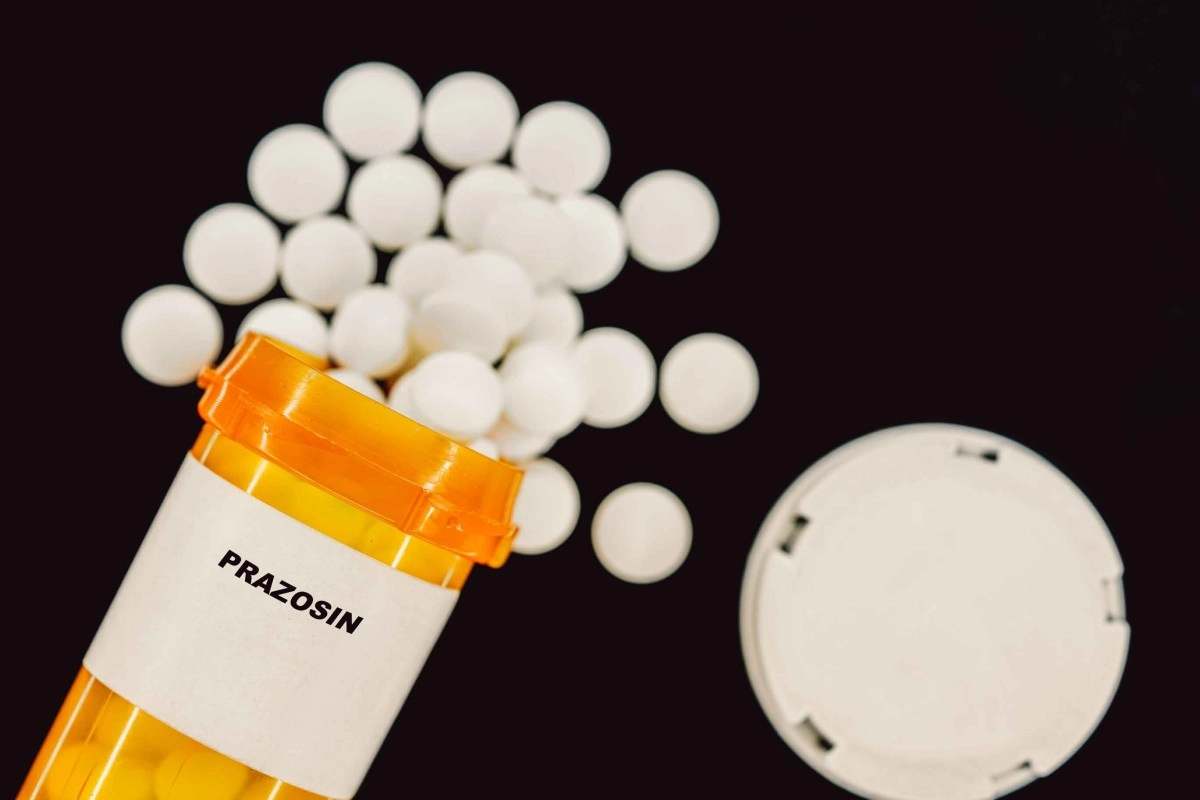
In a statement, the Food and Drug Administration (FDA) announced the recall of at least 580,000 bottles of prazosin hydrochloride, a blood pressure medication, which contains a substance that may cause cancer.
According to the health agency, the drug was manufactured by Teva Pharmaceuticals USA and distributed nationwide by Amerisource Health Services, who issued the alert after analyzing the drug. According to the FDA report, prazosin helps relax blood vessels and is used for high blood pressure, sleep disorders, and post-traumatic stress disorder. It is mostly available in 1mg, 2mg, and 5mg doses. On its website, the Food and Drug Administration stated that the recalled drug is classified as a Class II drug, explaining that it contains nitrosamine impurities considered potentially carcinogenic. Therefore, it requests that consumers discard the product or return it to the pharmacy where it was purchased for a refund.


